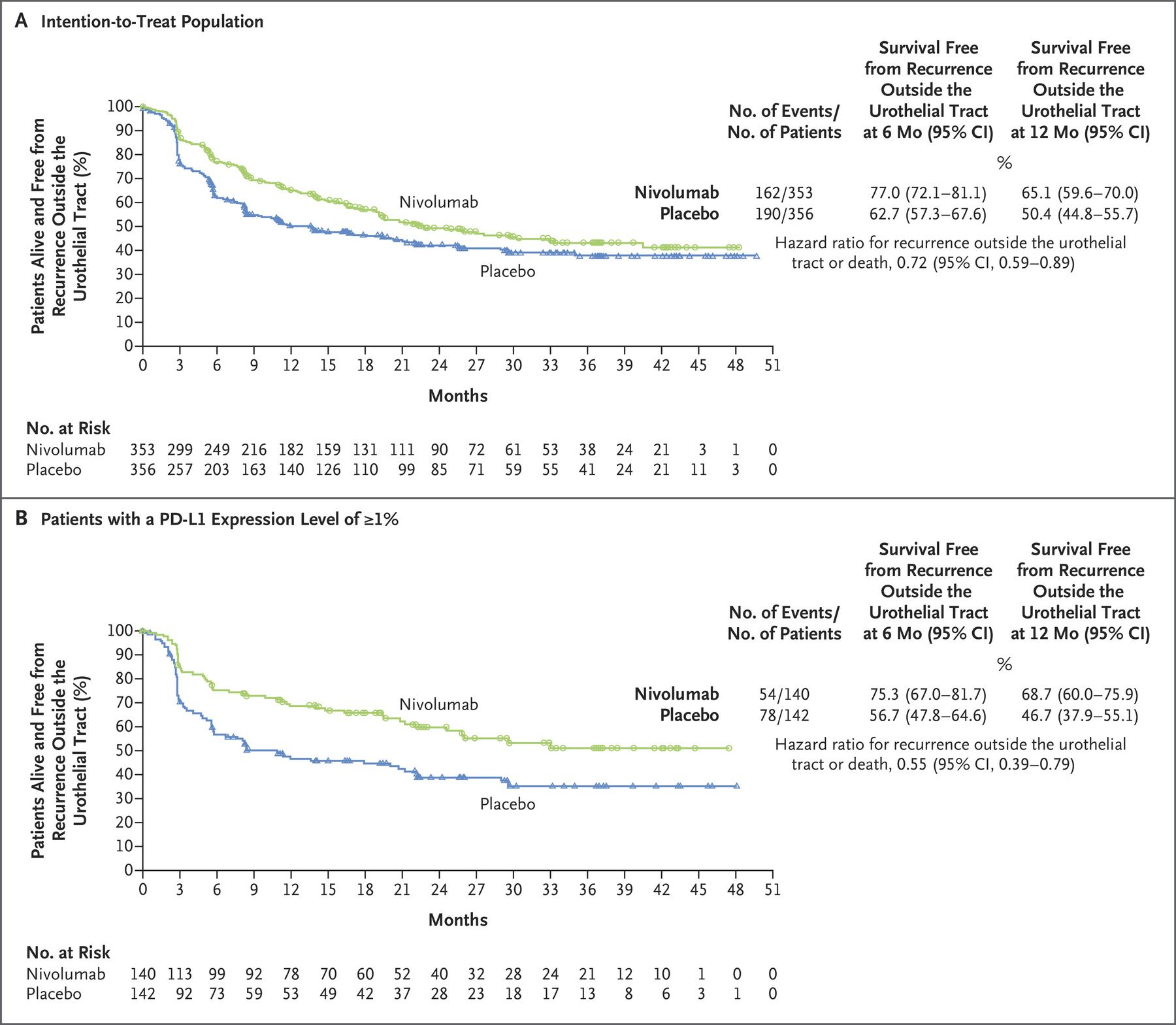
Increased Security: NTFS allows you to set permissions on a file or folder, specify the groups and users whose access you want to restrict or allow, and select access type.After a bad-sector error, NTFS dynamically remaps the cluster that contains the bad sector, making bad cluster and allocating a new cluster for the data. By using the log file and checkpoint information, it can restore the consistency of the file system (restore data) when the computer is restarted after a system failure. Increased Reliability: NTFS has a log file.Compared to them, NTFS has advantages including: NTFS is designed to replace FAT and HPFS. Starting with Windows NT 3.1 published in 1993, it is the default file system of the Windows NT family. NTFS (New Technology File System) is a proprietary journaling file system developed by Microsoft. Introduction to NTFS and ReFS What Is NTFS? Back up Server with MiniTool Partition Wizard.In this post, MiniTool Partition Wizard introduces ReFS vs NTFS to you and you can make a decision after reading this post. In November 2022, the drug was recommended by the National Institute of Health and Care Excellence (NICE) as a first-line treatment, alongside chemotherapy, for rare forms of advanced gastroesophageal cancer.Recently, many users are asking whether they should use ReFS or NTFS, for servers or large partition format. Opdivo is currently approved in more than 65 countries, including the US, EU, Japan and China for a range of indications such as melanoma, lung, kidney and gastric cancers. “We look forward to closely following the CheckMate -274 trial, which is ongoing to assess additional key secondary endpoints, including overall survival to which we currently remain blinded.” The latest results from BMS’ CheckMate-274 trial demonstrate significant sustained clinical benefits with Opdivo for the adjuvant treatment of patients with muscle-invasive urothelial carcinoma at a high risk of recurrence after radical resection.Īt a median follow-up of 36.1 months, Opdivo more than doubled the average length of time patients lived without disease recurrence, demonstrating a median disease-free survival of 22 months compared to 10.9 months with placebo.Īdditionally, those treated with Opdivo showed a median non-urothelial tract recurrence-free survival (NUTRFS) of 25.9 months compared to 13.7 months for placebo and, in patients whose tumour cells expressed PD-L1 of 1% and over, median NUTRFS was 52.6 months with Opdivo versus 8.4 months with placebo.īenefits were also seen across exploratory endpoints, including distant metastasis-free survival and second progression-free survival (defined as the time from randomisation to disease progression after subsequent next-line systemic therapy, start of second subsequent next-line systemic therapy or death).ĭana Walker, vice president, development programme lead, genitourinary cancers at BMS, said: “The durable follow-up results from CheckMate -274 continue to fuel our excitement toward our ongoing research in earlier stages and its potential to change outcomes for patients. The majority of urothelial carcinomas are diagnosed at an early stage, but rates of recurrence and disease progression are high and around 50% of patients who undergo radical surgery will experience disease recurrence. Urothelial carcinoma, which most frequently begins in the cells that line the inside of the bladder, accounts for approximately 90% of bladder cancer cases. Bristol Myers Squibb (BMS) has reported positive three-year follow-up results from its phase 3 trial of Opdivo (nivolumab) in certain patients with urothelial carcinoma, the most common type of bladder cancer.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
